Textbook Disinformation: “Weather Studies”

As astonishing as it may sound, we are witnessing a full-blown disinformation/re-education campaign against the children of the United States. They are being taught that chemical aerosols (geoengineering/”chemtrails”) being sprayed from jets are “harmless water vapor”.</p>GlobalSkywatch
This disinformation campaign poses profound implications.
First, it clearly demonstrates that the government fully intends to continue spraying for a long time. They are conditioning your children to believe that these chemical/metallic aerosols are “harmless” so they won’t protest, resist, or otherwise oppose the spraying when they grow up.
Second, a disinformation campaign of this scale reveals the alarming reality that we are witnessing the implementation of a plan that was set in motion years ago. This plan—to kill off a significant portion of the population—is being sold to the public as being for “the greater good of mankind”.
“Eugenic goals are most likely to be attained under another name than eugenics.” — Frederick Henry Osborn, 1937; Popular eugenicist, founding member of the American Eugenics Society, selected by Franklin Roosevelt to chair the Civilian Advisory Committee on Selective Service in 1940.
Why are America’s children being taught lies which cause them to accept the systematic poisoning of all life in Earth? Is it because powerful proponents of global depopulation serve in the most powerful political positions in the world?
Haven’t we seen this before? Do we really want to see it again?
How these questions are answered is entirely up to you.
“Weather Studies” Textbook
 This textbook is being used to teach children that chemical aerosols are harmless water vapor “condensation trails”, but this is blatantly false. Jet engines do not produce condensation trails except under extremely rare conditions, and these will be too short and too high to be seen from the group. In fact, most people will never see a true condensation trail in their entire lives. Many skywatchers and airline pilots have come forward to express their disdain about the blatant disinformation being told to the public regarding contrail formation.
This textbook is being used to teach children that chemical aerosols are harmless water vapor “condensation trails”, but this is blatantly false. Jet engines do not produce condensation trails except under extremely rare conditions, and these will be too short and too high to be seen from the group. In fact, most people will never see a true condensation trail in their entire lives. Many skywatchers and airline pilots have come forward to express their disdain about the blatant disinformation being told to the public regarding contrail formation.
Below is an excerpt from the school textbook entitled “Weather Studies“. We have provided photos and a transcript of chapter 7 which is entitled “Clouds, Prescription, and Weather Radar”.




Weather Studies
Chapter 7: “Clouds, Prescription, and Weather Radar”
CHAPTER 7
CLOUDS, PRECIPITATION, AND WEATHER RADAR
Case in Point
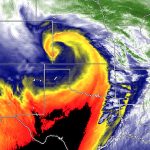
A familiar sight in the daytime sky almost anywhere is contrails, bright white streamers of ice crystals that form in the exhaust of jet aircraft (Figure 7.1). Contrails are modifying the cloud cover along heavily traveled air corridors between major urban areas with possible implications for weather and climate. According to some studies, contrails already cover about 5% of the sky in some portions of the eastern United States. A contrail, short for condensation trail, develops when the hot humid air in jet engine exhaust mixes with the cold, drier air at high altitudes. Turbulence in the exhaust causes the mixing. A similar process enables us to “see our breath” when we exhale on a cold winter day. For details on this process, see this chapter’s first Essay, “Clouds by Mixing.”
Depending on atmospheric conditions, contrails may dissipate (sublimate) within minutes or hours, or they may spread laterally forming wispy cirrus clouds that persist for a day or so. Atmospheric scientists hypothesized that increased cloud cover caused by contrails would effect the local radiation budget in two ways: During daylight hours, contrails and their cirrus byproducts reflect ?? incoming solar radiation thereby cooling Earth’s surface. At night, contrails and associated cirrus clouds absorb and emit infrared radiation welling up from below thereby enhancing the greenhouse effect and elevating Earth’s surface temperature. Cooler days and warmer nights reduces the diurnal temperature range (DTR), the difference between the day’s maximum and minimum temperatures. Furthermore, contrails may stimulate precipitation locally by supplying ice crystal nuclei for lower clouds.
Scientists had an opportunity to test their hypothesis regarding the impact of contrails on the radiation budget immediately after the terrorist attacks of 11 September 2001 when the Federal Aviation Administration (FAA) ordered a three-day shutdown of all commercial air traffic in the United States. Almost immediately, contrails began to dissipate. David J. Travis, a climate scientist at the University of Wisconsin-Whitewater, and colleagues at Pennsylvania State University analyzed the effects of a contrail-free sky on surface temperatures at 4,000 U.S. weather stations. They found that the DTR during the three-day period was about 1.1-Celsius degree (2 Fahrenheit degrees) higher than the long-term (30-year) average. Furthermore, the DTR for the three-day periods preceding and following the shutdown was below the long-term average.
These observations support the hypothesis that contrails may contribute to climate change by making days colder and nights warmer. With the expected future increase in global aircraft traffic, some scientists expect the contribution of contrails to climate change to become more significant; the average contrail sky coverage expected to more than double by 2050. At this point, it is not known whether more contrails will have a net warming or cooling effect on global climate.
Driving Question:
How do clouds and precipitation form!
Clouds whisk across the sky in ever-changing patterns of white and gray; fog lends an eerie silence to a dreary day. Clouds and fog are products of condensation or deposition of water vapor within the atmosphere. Most clouds are the consequence of saturation brought about by uplift and expansional cooling of air. Fog is a cloud that forms near, or in contact, with Earth’s surface; most fogs develop when the lowest layer of air is chilled to saturation via radiational or advective cooling.
Cloud formation is an essential part of the global water cycle because without clouds there would be no rain or snow. And yet most clouds—even those associated with large storm systems—do not produce precipitation. The principal subjects of this chapter are the development and classification of clouds and fog, plus the formation and types of precipitation. We also examine weather radar, a valuable tool for locating and tracking areas of precipitation and for monitoring air motion within weather systems. This chapter closes with a summary of the various direct and remote methods for measuring precipitation.
Cloud Formation
Water vapor is an invisible component of air, but its condensation and deposition products (water droplets and ice crystals) are visible. A cloud is the visible product of condensation or deposition of water vapor within the atmosphere; it consists of an aggregate of minute water droplets and/or ice crystals suspended in the atmosphere. This section covers the process of cloud development.
Clouds are increasingly likely to form as air nears saturation. But when scientists try to simulate this process in the laboratory using a clean-air chamber, a cloud does not form even if the relative humidity is elevated well above 100%, that is, to supersaturation values. A clean-air chamber is an enclosed container of air from which all solid and liquid particles (aerosols) have been filtered out. Introducing water vapor into the chamber raises the relative humidity, but even if the relative humidity is elevated to as high as 200%, no cloud forms. How is that possible?
We expect that once air becomes saturated (relative to a flat-water surface), condensation would initially produce extremely small droplets and those droplets would eventually grow into cloud droplets through additional condensation. However, droplets will not form in the first place unless the relative humidity rises to extraordinarily high levels of supersaturation.
THE CURVATURE EFFECT
Tiny droplets cannot form in a cloud chamber without a supersaturated environment because of the so-called curvature effect. The curvature of a water surface affects the ability of water molecules to escape (vaporize) from the water surface. The smaller the droplet, the greater is the concentration of surrounding water vapor that is necessary for the droplet to grow. The curvature of the surface of a spherical water droplet increases as the radius of the droplet decreases; that is, the surface of a small droplet has a greater curvature than the surface of a large droplet. (At the limit, a flat-water surface has no curvature.) With increasing curvature, molecules that form the surface of a droplet have fewer neighboring molecules and are more weakly bonded. Molecules composing a flat-water surface have the most neighboring molecules and are most strongly bonded. Bond strength affects the flux of water molecules from the liquid to vapor phase. For this reason, water molecules more readily escape a water droplet than a flat-water surface and water molecules more readily escape a small water droplet than a large water droplet. Consequently, at the same temperature, the saturation vapor pressure surrounding a small water droplet is greater than that surrounding a large droplet.
Values of saturation vapor pressure listed in Table 6.3 are defined for air over a flat surface of fresh water and are the basis for computing relative humidity (Chapter 6). The saturation vapor pressure increases with increasing curvature (and decreasing droplet size). For droplets having a radius of 0.001 micrometer, the saturation vapor pressure at a given temperature is about 3.4 times that specified in Table 6.3, which computes to a relative humidity of 340%. On the other hand, for droplets having a radius greater than 1.0 micrometer, the difference between the saturation vapor pressure surrounding the droplet and the values listed in Table 6.3 is minimal and computes to a relative humidity only slightly higher than 100%.
The bottom line: In clean air, water vapor will not condense into tiny water droplets unless the air is supersaturated to very high levels. In the real atmosphere, the relative humidity never even approaches such great supersaturation values, so tiny droplets cannot form or grow. What then makes cloud formation possible?
ROLE OF NUCLEI
The clean-air chamber experiment described above failed to produce a cloud because the air was not sufficiently supersaturated. In the real atmosphere, supersaturated air is not needed for clouds to develop; in fact, cloud droplets readily form as the relative humidity nears 100%. And clouds are abundant; at any given time, about 60% of the planet is shrouded in clouds. How then, do cloud droplets form? The answer: they have a head start. Suspended in air are solid and liquid particles that provide relatively large surface areas on which condensation or deposition initially takes place. These naturally occurring particles that promote condensation or deposition in the atmosphere are known as nuclei. Nuclei have radii greater than 1.0 micrometer and favor condensation (or deposition) of water vapor. Once condensation (or deposition) starts, they become comparably sized cloud droplets (or ice crystals). Additional growth of these droplets (or ice crystals) by condensation (or deposition) is more likely because they can grow in an environment with a relative humidity that exceeds 100% by only 1% or so. Most cloud particles have radii of 2 to 10 micrometers.
Nuclei, essential for cloud formation, are abundant in the atmosphere and are continually cycled into the atmosphere from Earth’s surface. Sources of nuclei include volcanic eruptions, wind erosion of soil, forest fires, and ocean spray. When sea waves break, drops of salt water enter the atmosphere and the water evaporates leaving behind tiny sea-salt crystals that function as nuclei (Figure 7.2). Emissions from domestic and industrial chimneys also contribute nuclei to the atmosphere.
Depending on the product (liquid water droplets or ice crystals), a distinction is made between cloud condensation nuclei and ice-forming nuclei. Cloud condensation nuclei (CCN) promote condensation of water vapor at temperatures both above and below the freezing point of water. Within the atmosphere, water vapor can condense into cloud droplets that remain liquid even at temperatures well below 00 C (320 F). Droplets at such temperatures are described as supercooled. Ice-forming nuclei (IN) are much less common than CCNs and promote formation of ice crystals only at temperatures well below freezing. The most efficient ice-forming nuclei are substances having a crystal structure similar to that of ice and most are almost insoluble in water. Clayey soil particles are excellent ice-forming nuclei. The two types of ice-forming nuclei are freezing nuclei and deposition nuclei. Freezing nuclei are particles on which water vapor condenses and subsequently freezes; they are active at temperatures below about -90 C (160 F).
Hygroscopic nuclei are a special category of cloud condensation nuclei because they possess a chemical attraction for water molecules. Condensation begins on hygroscopic nuclei at a relative humidity under 100%. Magnesium chloride (MgCl2), a salt in sea-spray, can promote condensation at a relative humidity as low as 70%! Clouds form more efficiently where hygroscopic nuclei are abundant. Many sources of hygroscopic nuclei exist in urban-industrial areas and this helps explain why localities downwind of large cities tend to be somewhat cloudier and rainier than upwind localities. The Metropolitan Meteorological Experiment (METROMEX) conducted in the 1970s demonstrated that the average summer rainfall was 5% to 25% greater within, and up to 50 to 75 km (31 to 47 mi) downwind of St. Louis, MO than upwind of the city. In more recent years, similar urban effects on precipitation were detected in Tokyo, Phoenix, and other cities. Besides being a source of hygroscopic nuclei, cities also spur cloud and precipitation development by contributing water vapor (raising the relative humidity) and heat (adding to the buoyancy of air). Furthermore, the relative roughness of a city surface induces convergence of horizontal winds, uplift of air, and expansional cooling (Chapter 8).
Interestingly, not all ice crystals form on the traditional nuclei described above. While sampling ice crystals in cold clouds from aircraft, scientists discovered that the concentration of ice crystals is highly variable within the cloud and ice crystals are 10 to 1,000 times more numerous than nuclei. This discrepancy is likely explained by the presence of fragments of ice crystals that break off during midair collisions. These ice fragments then function as additional nuclei.





























































































































I am ashamed that these lies will be taught to our next generation. I think it is important to start educating your children in everyday conversation about the truth. We are there only hope for the truth. They will never learn the truth in the schools, the media or from our government officials. I have even spoken to my 5 year old about the chemtrails. She is at the age where she will maybe not fully understand but kids absorb more than we think. That is all we can do about is educate those around us.. I don’t have any problems telling my children the truth despite what they have learned at school. Older children who are able to get on the computer can easily be directed at resources to validate what you are teaching them. Show them the videos “What in the world are they spraying,” and “Why in the world are they spraying.” Thank you Dane for all your dedication and relentless drive to educate people. It is not easy when you have so many folks who reject the idea of chemtrails and geoengineering.
God Bless.
We are fighting back in the schools.
http://www.youtube.com/watch?v=Qrf5pF3SRKk
Thank you! https://www.geoengineeringwatch.org/student-fights-back-against-disinformation/
once i got to the part where they tell the little slaves that these “contrails” cover up to five percent of the sky and can last minutes or hours i did not want to continue and waste minutes of my life listening to these blatant lies. i would just like to tell you Dane i appreciate all that you do and i pray for you and your familys well being. stay safe and know that its possible other technologies could be being used on the population that resist and disementate information. I was hoping you could forward me some data with advice on how to raise children in this matrix we live in. thank again, namaste. “just victims of the in house drive-by, they say jump and you say, how high”!
Zack from Rage against the Machine.
[…] [More…] […]
I confirm exactly what John has described. It was perfectly clear skies over Toronto for 2-3 days even though air traffic to and from Pearson airport was the same daily average, followed by spraying on 27-May with an intensity not noticed before.
Well, jeez, “Larry”, the phenomena is beyond dispute. As an ex pilot I have seriously been observing geoengineering (aka Chem Trails) over Toronto for some time now. We just had a relatively clear period for over a week, corresponding with an extended and coordinated Memorial period stand down (even military pilots need a rest), then the sky was ripped up May 27 with smoke trails criss crossing the skies, large X appearing, at higher altitudes easily observed as tanker like large aircraft (modified DC-10). And total and complete cover up by mainstream media. Why isn’t this phenomena being reported by media, because it is dangerous for them to report it so. Conditioning our children will clearly be part of their nightmarish agenda.
Seriously???